Soil pH - Acid Soil or Alkaline Soil - What You Need to Know
What is Soil pH?
It is a measure of the number of H+ ions in the soil water. pH means potential Hydrogen.
Are you glad I cleared that up for you? Or is it just about as clear as mud?
For us lay people it's enough to know that Soil pH is a measure of how acidic or alkaline our soil is.
The pH is a logarithmic scale that goes from 0 to 14 with 0 being most acidic, 7 being neutral and 14 being most alkaline. Because it's a logarithmic scale each jump is a ten fold increase so a soil with a pH of 5 is 10 times more acidic than one with a pH of 6 and a 100 times more acidic than one with a pH of 7.
Your Soil pH can have a profound effect on your garden especially for those of you who rely on chemical fertilizers. It will have a significant effect what nutrients are available to your plants.
Are you already using compost and working more closely with the soil microorganisms? If so you will soon find those critters compost brings to your soil working their magic, buffering pH levels to suit the plants you are growing.
What Causes Soil to Become Acidic or Alkaline?
A big part of soil pH is intrinsic to the soil and the climate where you live. In other words if you have a very high or very low pH reading for your soil, it isn't necessarily because of something you did or something you have a lot of control over. The main causes are:
- Parent Material - Soils are made up of ground up rock and the type of rock dictates the natural pH of the soil. Basic rock like limestone creates alkaline soil and acidic rock, rock containing more silica, creates acidic soil.
- Rainfall - Rain leaches basic elements such as calcium and magnesium from the soil. Therefore you will find areas with high rainfall generally have acidic soil while dry areas tend to have alkaline soil. In fact the work for deserts in Arabic is al khali.
- Fertilizers - Some fertilizers tend to make the soil acidic. Ammonium urea is an example.
How Does Soil pH Affect Plants?
Most plants like a soil pH close to neutral or just a little on the acid side. A very high or very low pH can be toxic to the plants.
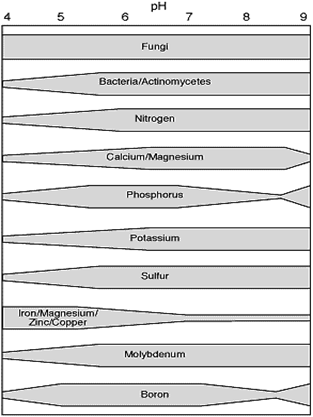
This graphic shows how soil pH affects the usual level of availability of some of the nutrients your plants need. It also shows how some of the important members of the soil biological community respond to different pH levels.
In Acid soils calcium (Ca), and magnesium (Mg) are less available to plants. Aluminum (Al) and manganese (Mn) may reach toxic levels. Phosphorus is tied up by iron (Fe) and aluminum (Al). Bacteria grow poorly as well.
In Alkaline soils phosphorus (P) gets tied up by Ca and Mg. Iron (Fe), zinc (Zn) and manganese (Mn) are less available. Sometimes excess salts accumulate and potatoes are subject to bacterial diseases.
Have you noticed that Phosphorus is too fussy. It's not available if the soil is acidic and it's not available when the soil is alkaline. So when is phosphorus available?
Phosphorus is a critical nutrient, one of the big three. However, most additions of phosphorus fertilizers, whether synthetic or organic, tend to get tied up in the soil.
So how the heck do plants get their phosphorus? Most plants form a relationship with mycorrhizal fungi. As you can see fungi tend to function through all ranges of pH. One of their most important functions is solubilizing phosphorus and channeling it to plants. In turn plants feed the fungi sugars. Check this page on phosphorus and fungi to learn more.
How Does Compost Help?
Compost is useful to add to soils regardless of whether you have acidic or alkaline soil. How can it do this? Compost is a buffering agent.
A buffer is a substance that tends to lessen the change in pH. Compost allows the soil pH to effectively range to one point above and below the pH reading for your soil. So for example, I have a pH of 8. Adding compost gives me a range of 7 to 9 pH. As 7 is in the ideal range for plants I've solved a good part of my pH problem with the compost.
Compost also inoculates the soil with a whole range of soil microbes. The organic matter both provides a habitat for the bacteria, fungi and others to make their homes and feeds them.
The most intense population of soil critters will be found right next to the roots and the mycorrhizal fungi attached to those roots. In healthy biologically active soil there will be a very diverse community of soil microbes feeding plants nutrients and being fed by the sugars the plants produces called exudates.
This community affects the soil pH, tending to neutralize pH levels in the rhizosphere. I have to check this out this summer with a few extra pH tests of the soils attached to the roots.
How to Raise the pH of Acidic Soils
As your first step try compost. It can have a marked influence on acid soils. For example an EPA study showed that water with a pH of about 2 discharged from a mine had risen to a pH of 5 by the time it passed through a compost filter.
Go slowly. It is possible to use quick acting agents to radically change your soil pH. However, you risk wiping out your soil organisms and they are critical companions to your plants. Kill them and you effectively kill your soil.
Aim for raising your pH a maximum of 1 pH point per season. Lime is normally added to raise pH. You can use Calcitic or Dolomite Lime. Calcitic Lime adds only calcium to your soil while Dolomite lime adds calcium and magnesium. These will slowly change the pH. As well it will make phosphorus more available and increase the bacterial activity with the side effect of freeing nitrogen for your crop.
The amount you add depends on your soil texture. In sandy soils use less lime and apply it more often. Follow the directions on the lime you purchase but please go slow - give soil life a chance.
How to Lower the Soil pH of Alkaline Soils
I've found that many gardening sites and books throw up their hands in defeat when faced with soil pH in the alkaline range. Personally I think it's that there are fewer people here in the dry areas where soils tend to be basic. These garden experts are not terribly motivated to solve this problem because they don't happen to have any experience of it. So they simply write soils with high pH off as impossible.
The other thing - and this concerns me - is that because lime is so routinely applied to soils in the heavily populated areas where soils are acidic you find lime for sale in garden centers where soils are alkaline. The other day I was at a big garden center in Calgary, an alkaline area, and found a huge display of bags of lime for sale. Many people from acid climes are moving here and naturally think their garden problems will be solved with lime. In fact they will be made a heck of a lot worse.
So - please do your soil tests before you try to fix your soil and then go slow in effecting changes. Give your soil biology a chance to adjust and adapt to the new conditions.
Your first line of action is compost. It does work to change the actual pH and to minimize the difficulties arising from a high pH.
To lower your soil's pH sulfur is your friend. It takes very little but it takes some time. The equation is sulfur + time + soil bacteria = a lowered pH. Again you will need less sulfur in sandy soils than in other soils so know your soil texture. Follow the directions that come with the material you purchase and don't try to change things more than one pH point per season.
How to Test Your Soil pH - Here are three different ways to test your soil pH.
Lowering Your Soil pH - Those of you living in areas with alkaline soils check out this page.
Happy soil explorations people!